
Antioxidant BHT 264
CAS:128-37-0
Purity:99%
Contact Now
We will contact you as soon as possible
Your Location:Home >Products >Organic Chemistry >1493-13-6
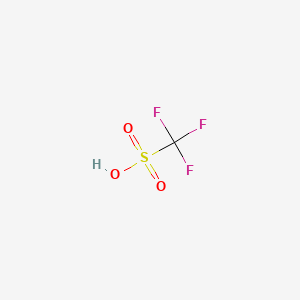

Product Details
|
Chemical Description |
Trifluoromethanesulfonic acid is an organic compound with the formula CF3SO3H. |
|
|
|
|
Preparation |
Yellow-brown liquid. The boiling point is 167~170 ℃.The refractive index is 1.331.The relative density is 1.708.It is the strongest organic acids, easily soluble in water.Use carbon disulfide as raw material, with the reaction of iodine pentafluoride to produce trifluoromethyl disulfide.(CF3S) 2Hg was obtained when reacting with mercury; Then through oxidation of hydrogen oxide, trifluoromethanesulfonic acid is acquired. |
|
Reactions |
Trifluoromethanesulfonic acid acts as a catalyst for esterification reactions and an acidic titrant in nonaqueous acid-base titration. It is useful in protonations due to the presence of conjugate base triflate is non nucleophilic. It serves as a deglycosylation agent for glycoproteins. In addition, it is a precursor and a catalyst in organic chemistry. It reacts with acyl halides to prepare mixed triflate anhydrides, which are strong acylating agents used in Friedel-Crafts reactions. It acts as a key starting material for the preparation of ethers and olefins by reacting with alcohols as well as to prepare trifluoromethanesulfonic anhydride by dehydration reaction. Catalyst used in the production of cocoa butter substitute from palm oil. This is a very similar reaction to what would be done if one wanted to create polymers using triflic acid in the synthesis. Other Friedel-Crafts type reactions using triflic acid include cracking of alkanes and alkylation of alkenes which are very important to the petroleum industry. These triflic acid derivative catalysts are very effective in isomerizing straight chain or slightly branched hydrocarbons that can increase the octane rating of a particular petroleum based fuel. |
|
Definition |
ChEBI: Trifluoromethanesulfonic acid is a one-carbon compound that is methanesulfonic acid in which the hydrogens attached to the methyl carbon have been replaced by fluorines. It is a one-carbon compound and a perfluoroalkanesulfonic acid. It is a conjugate acid of a triflate. |
|
General Description |
Trifluoromethanesulfonic acid is a strong organic acid. It can be prepared by reacting bis(trifluoromethylthio)mercury with H2O2. On mixing with HNO3, it affords a nitrating reagent (a nitronium salt). This reagent is useful for the nitration of aromatic compounds. Its dissociation in various organic solvents has been studied. |
|
Safety Profile |
A corrosive irritant to the skin, eyes, and mucous membranes. A strong acid. Violent reaction with acyl chlorides or aromatic hydrocarbons evolves toxic hydrogen chloride gas. When heated to decomposition it emits toxic fumes of Fand SOx. See also FLUORIDES. |
InChI:InChI=1/CHF3.H2O3S/c2-1(3)4;1-4(2)3/h1H;4H,(H,1,2,3)
Perfluoroalkyl perfluoroalkanesulfonates...
The Raman and i.r. spectra of trifluorom...
ECF processes have been extensively expe...
Recently we reported a study of the ther...
A low-lying ?* level localized on a sulp...
The synthesis of the first trifluorometh...
Much enhanced acid catalysis was observe...
-
Trifluoromethane is transformed to trifl...
Water is found able to determine the sel...
The present invention concerns a method ...
The present invention relates to a proce...
trifluoromethanesulfonic acid ethyl ester

benzene

trifluorormethanesulfonic acid

ethylbenzene

ortho-diethylbenzene

9,10-dimethylanthracene
| Conditions | Yield |
|---|---|
|
|
methanol

4-tert-butylphenyl triflate

formic acid

tert-butylbenzene

Dimethoxymethane

dimethylsulfite

trifluoromethan

trifluorormethanesulfonic acid

para-tert-butylphenol

1-(tert-butyl)-4-methoxybenzene

hydrogen fluoride

sulfur dioxide
| Conditions | Yield |
|---|---|
|
With oxygen; for 6h; Reagent/catalyst; Mechanism; Quantum yield; Photolysis;
|
5 %Chromat. 60 %Chromat. |
trifluoromethanesulfonic acid ethyl ester
Trifluoromethanesulfonyl fluoride
diethyl ether
methyl trifluoromethanesulfonate
trifluoromethane sulfonyl chloride
trifluoromethylsulfonic anhydride
ethoxycarbonylmethyl trifluoromethanesulfonate
trifluoromethanesulfonic acid 2,2,2-trifluoro-1-phenyl-1-trifluoromethyl-ethyl ester